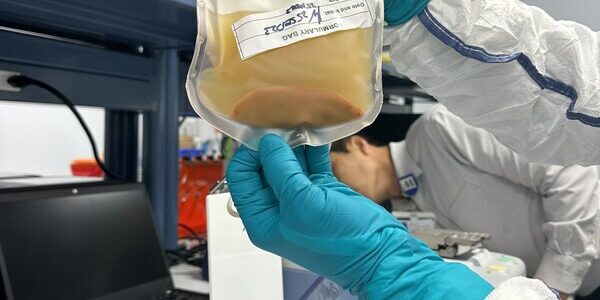
LyGenesis Announces First Patient Dosed in its Phase 2a Clinical Trial of a First-in-Class Regenerative Cell Therapy for Patients with End-Stage Liver Disease
LyGenesis, a clinical-stage biotechnology company developing cell therapies for large unmet medical needs, announced today that the first patient has been dosed in their Phase 2a clinical trial evaluating their first-in-class allogenic regenerative cell therapy transplanted into patients’ lymph nodes…
NeuroStar® Advanced Therapy Receives FDA Clearance as a First-Line Add-On Treatment for Adolescents with Depression
Neuronetics, Inc. (NASDAQ: STIM), a medical technology company focused on designing, developing, and marketing products that improve the quality of life for patients who suffer from neurohealth disorders, announced clearance from the U.S. Food and Drug Administration (FDA) for NeuroStar…
Madrigal Pharmaceuticals Announces FDA Approval of Rezdiffra™ (resmetirom) for the Treatment of Patients with Noncirrhotic Nonalcoholic Steatohepatitis (NASH) with Moderate to Advanced Liver Fibrosis
Madrigal Pharmaceuticals, Inc. (NASDAQ:MDGL), a biopharmaceutical company focused on delivering novel therapeutics for nonalcoholic steatohepatitis (NASH), today announced that the U.S. Food and Drug Administration (FDA) has granted accelerated approval for Rezdiffra (resmetirom) in conjunction with diet and exercise for…
Wegovy® receives FDA approval for cardiovascular risk reduction in adults with known heart disease and overweight or obesity
Novo Nordisk today announced that the U.S. Food and Drug Administration (FDA) has approved an additional indication for Wegovy® to reduce the risk of major cardiovascular events such as death, heart attack, or stroke in adults with known heart disease and…
Formosa Pharmaceuticals and AimMax Therapeutics Receive FDA Approval for Clobetasol Propionate Ophthalmic Suspension 0.05%, for the Treatment of Post-Operative Inflammation and Pain Following Ocular Surgery
Taiwan-based Formosa Pharmaceuticals (6838.TWO) and AimMax Therapeutics (United States) announced today that the U.S. Food and Drug Administration (FDA) has approved clobetasol propionate ophthalmic suspension 0.05% (APP13007), for the treatment of post-operative inflammation and pain following ocular surgery. Utilizing…
RYBREVANT® (amivantamab-vmjw) in Combination With Chemotherapy Is the First FDA Approved Therapy for First-line Treatment of Patients With Non-Small Cell Lung Cancer with EGFR Exon 20 Insertion Mutations
Johnson & Johnson (NYSE: JNJ) announced today that following a priority review, the U.S. Food and Drug Administration (FDA) has approved RYBREVANT® (amivantamab-vmjw) in combination with chemotherapy (carboplatin-pemetrexed) for the first-line treatment of patients with locally advanced or metastatic non-small cell lung…
Everest Medicines’ Partner Pfizer Announces European Commission Approves VELSIPITY® for Patients with Moderately to Severely Active Ulcerative Colitis
Everest Medicines (HKEX 1952.HK, “Everest”, or the “Company”)’s partner Pfizer Inc. (NYSE: PFE) announced that the European Commission (EC) has granted marketing authorization for VELSIPITY® (etrasimod) in the European Union to treat patients 16 years of age and older with moderately to…
Melanoma Research Alliance Applauds FDA Approval of First Cellular Therapy in Melanoma
The Melanoma Research Alliance (MRA), the largest non-profit funder of melanoma research worldwide, welcomes the U.S. Food and Drug Administration (FDA) decision to approve Iovance Biotherapeutics’ AMTAGVI™ (lifileucel) for the treatment of patients with advanced melanoma in the second line treatment setting….
Ipsen’s Onivyde® regimen, a potential new standard-of-care first-line therapy in metastatic pancreatic adenocarcinoma, approved by FDA 
Ipsen (Euronext: IPN; ADR: IPSEY) announced today that the U.S. Food and Drug Administration (FDA) has approved the supplemental new drug application for Onivyde® (irinotecan liposome injection) plus oxaliplatin, fluorouracil and leucovorin (NALIRIFOX) as a first-line treatment in adults living with…
Outlook Therapeutics® Doses First Subject in NORSE EIGHT
Outlook Therapeutics, Inc. (Nasdaq: OTLK), a biopharmaceutical company working to achieve FDA approval for the first ophthalmic formulation of bevacizumab for the treatment of retinal diseases, today announced that the first subject has been dosed in the NORSE EIGHT clinical…