Dupixent® (dupilumab) is now approved in India for the treatment of adults with moderate-to-severe atopic dermatitis
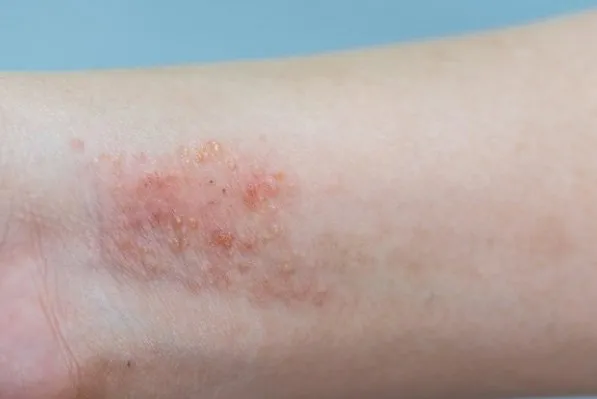
Sanofi Healthcare India Pvt. Ltd. has announced a milestone in dermatological treatment with the approval of Dupixent® (dupilumab), a novel biologic medicine for adults with moderate-to-severe atopic dermatitis. Unlike traditional treatments that broadly suppress the immune system, Dupixent® specifically targets the type 2 inflammation that underlies the disease, revolutionizing the approach to treatment.
Atopic dermatitis, a form of eczema, is a chronic type 2 inflammatory disease. Its manifestations include persistent intense itching, skin dryness, redness, and oozing. These symptoms often cause significant distress, leading to a considerable impact on patients’ quality of life, including disrupted sleep, anxiety, depression, and social stigma.
The introduction of Dupixent® marks a significant shift in atopic dermatitis management in India. Already approved in over 60 countries, including the U.S., the European Union, and Japan, Dupixent® has demonstrated significant improvements in disease symptoms and quality of life for patients with this difficult-to-treat skin condition.
Developed under a global collaboration between Sanofi and Regeneron, Dupixent® has been administered to over 600,000 patients worldwide. With this recent approval, it is set to impact the Indian medical landscape positively, offering a much-needed treatment alternative for adults suffering from moderate to severe atopic dermatitis.