AbbVie Employees in Mumbai Volunteer During 9th Annual “Week of Possibilities” to Support Young Adults with Cerebral Palsy
AbbVie, a global biopharmaceutical company focused on creating innovative medicines and solutions that address complex health issues and enhance people’s lives, announced the ninth annual ‘Week of Possibilities.’ It is a global volunteering program that unites employees with a shared…
Experts Highlighted the Significance of Collaborative Healthcare Efforts Between India & Nigeria
In a world reshaping after the pandemic, healthcare has taken centre stage. Recently, the inaugural session of HEAL-Thy Samvad brought together healthcare experts, policymakers, and a representative from Nigeria, a key partner in the Global South, to explore collaborative approaches…
Exactech Enhances High Technology Knee System with New Balancing Software
Exactech, a global medical technology leader, unveiled the next evolution of its ligament-driven balancing technology with new ExactechGPS® software that accommodates patient-centric planning and modern alignment philosophies for total knee replacement surgery. The software release upgrades the company’s proprietary Newton® Knee balancing technique to…
Introducing Ignians’ Total Multivitamin & Sleep Aid Capsules: The Ultimate Solution for High Performers
As the demands of modern life continue to increase, high performers in various industries understand the crucial connection between health and peak performance. Today, Ignians Inc. proudly unveils their latest innovation: Total Multivitamin & Sleep Aid Capsules, designed specifically for…
4 best health supplements to promote overall wellness
Now forget the basic healthcare routine – Introducing cutting-edge supplements designed to support your overall health from within. Starting from your skin, internal health and sexual health, these innovative formulas provide essentials that your body needs to enhance overall wellness….
SMBXL elevates the digital experience for MSMEs by introducing India’s largest B2B and B2C Online Hospital and Medical Supplies Expo
SMBXL, a leading technology company that supports MSMEs, has launched India’s largest ONLINE B2B and B2C Hospital & Medical Supplies Expo. The expo has over 1200+ companies from the Hospital & Medical Supplies sector across India. The participating organisations represent…
DRAWBRIDGE SECURES FDA 510(K) CLEARANCE FOR AT-HOME BLOOD SAMPLING DEVICE NANODROP
Drawbridge Health, a healthcare technology company focused on reinventing the blood draw experience, today announced that its at-home, patented blood sampling device, NanoDrop, has received U.S. Food and Drug Administration (FDA) 510(k) clearance for Over-the-Counter use. The clearance of this…
Mars Veterinary Health pilots industry-leading anaesthetic gas capture technology to reduce carbon emissions
Mars Veterinary Health, through its UK veterinary group Linnaeus and in partnership with Waltham Petcare Science Institute, today announced a first-of-its-kind partnership with SageTech Veterinary to pilot an innovative anaesthetic gas capture solution to help prevent harmful greenhouse gases from entering the atmosphere. During a…
SweetWater Brewing Launches Hammer Red Amber Ale to Support Georgia Aquarium
SweetWater Brewing Company (“SweetWater Brewing” or “SweetWater”), the largest craft brewer in the Southeast and a subsidiary of Tilray Brands, Inc. (NASDAQ: TLRY and TSX: TLRY), announced today the release of its new Hammer Red Amber Ale, the latest collaboration beer as…
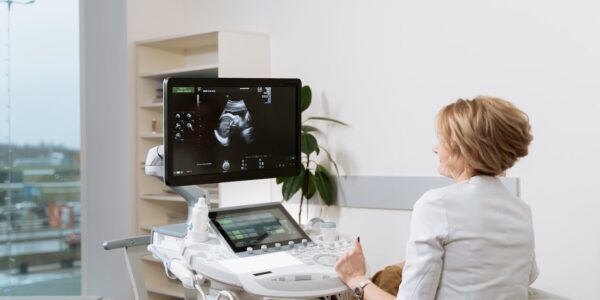
SmartAlpha Partners with Siemens Healthineers to advance AI-powered Workflow Efficiencies in Ultrasound
SmartAlpha has announced a partnership with Siemens Healthineers with a signing ceremony at the European Congress of Radiology (ECR). This partnership signifies a milestone, by both companies, to provide innovative AI-powered applications that improve workflow efficiencies with consistency to optimize clinical…